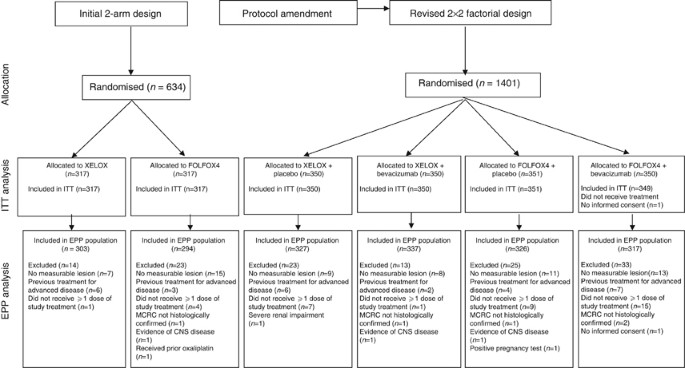
XELOX vs FOLFOX-4 as first-line therapy for metastatic colorectal cancer: NO16966 updated results | British Journal of Cancer

The prognostic effect of adjuvant chemotherapy in the colon cancer patients with solitary lymph node metastasis | International Journal of Colorectal Disease

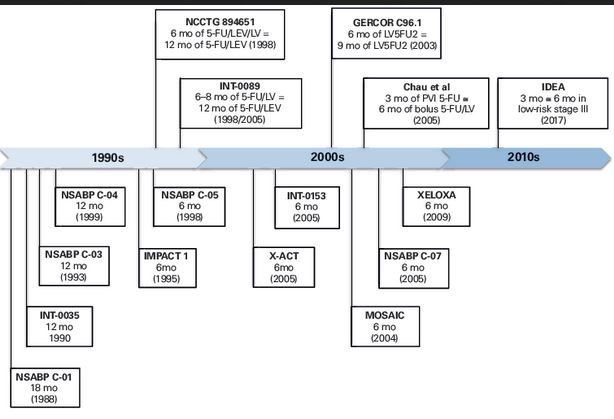
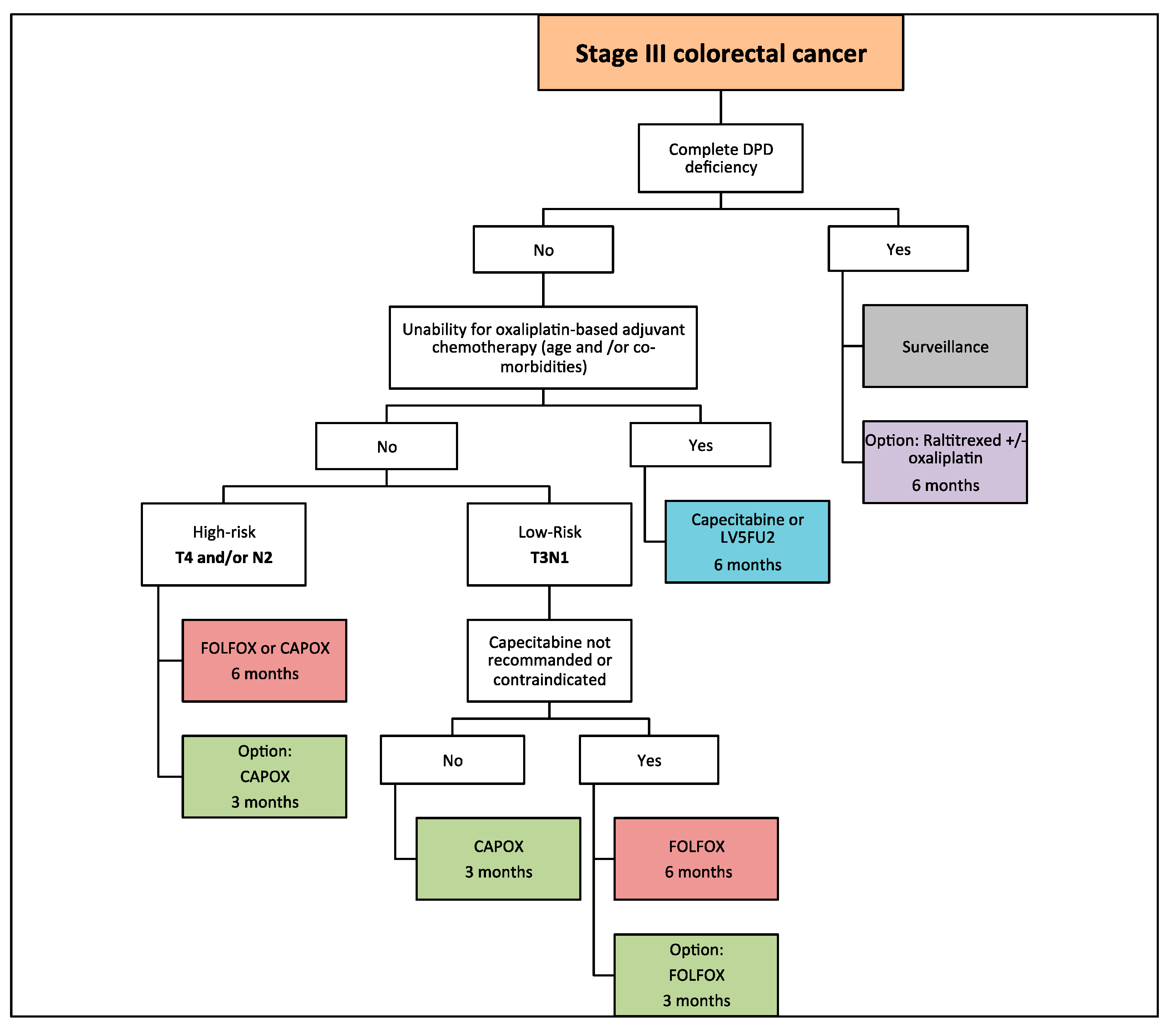
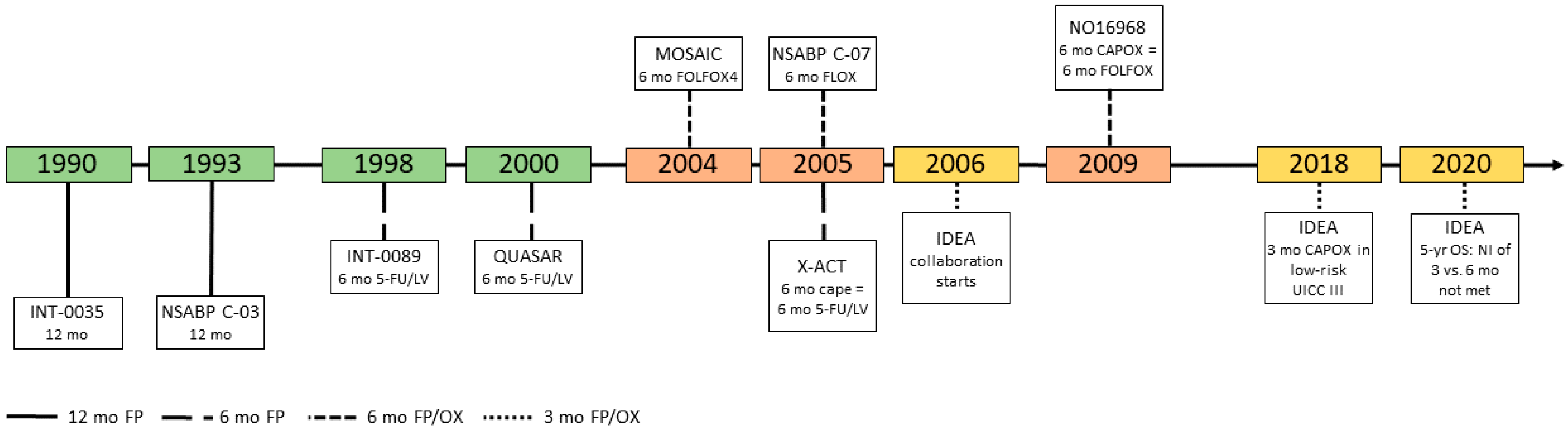
3. Cancer du côlon non métastatique | SNFGE.org - Société savante médicale française d'hépato-gastroentérologie et d'oncologie digestive

Median cycle of XELOX adjuvant chemotherapy received by patients for... | Download Scientific Diagram

JCM | Free Full-Text | The Role of Prophylactic and Adjuvant Hyperthermic Intraperitoneal Chemotherapy (HIPEC) in Prevention of Peritoneal Metastases in Advanced Colorectal Cancer
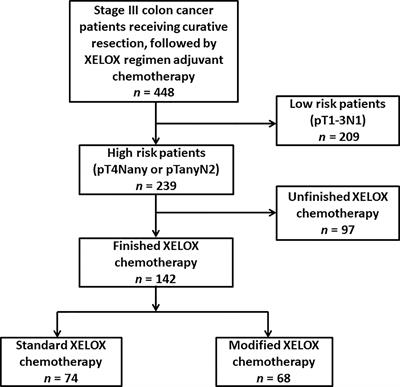
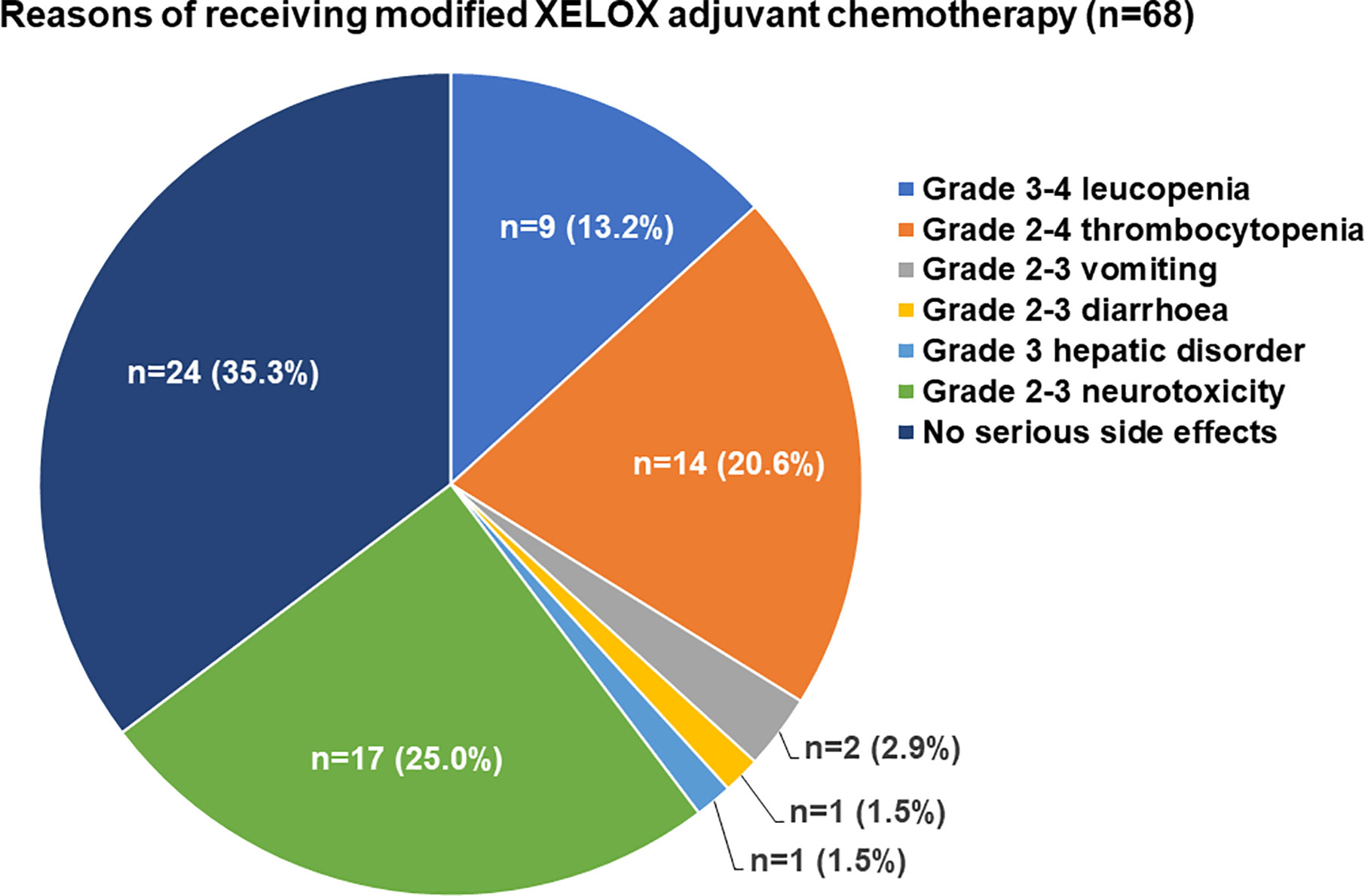
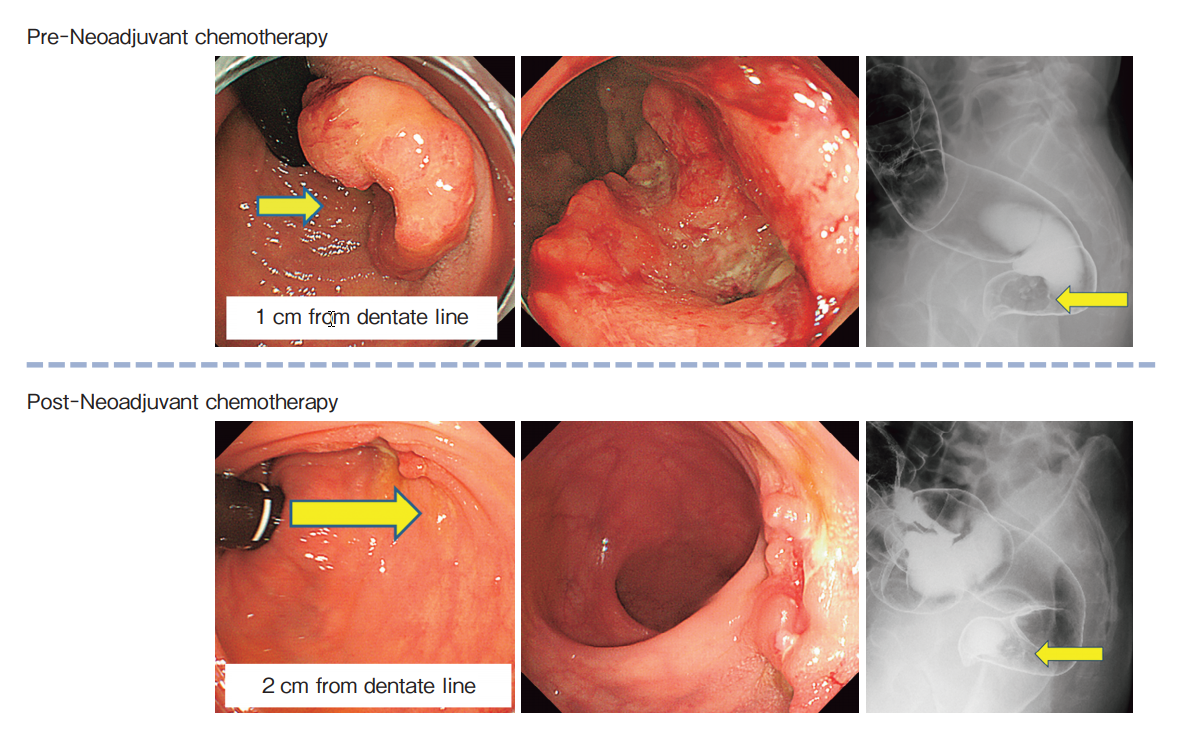
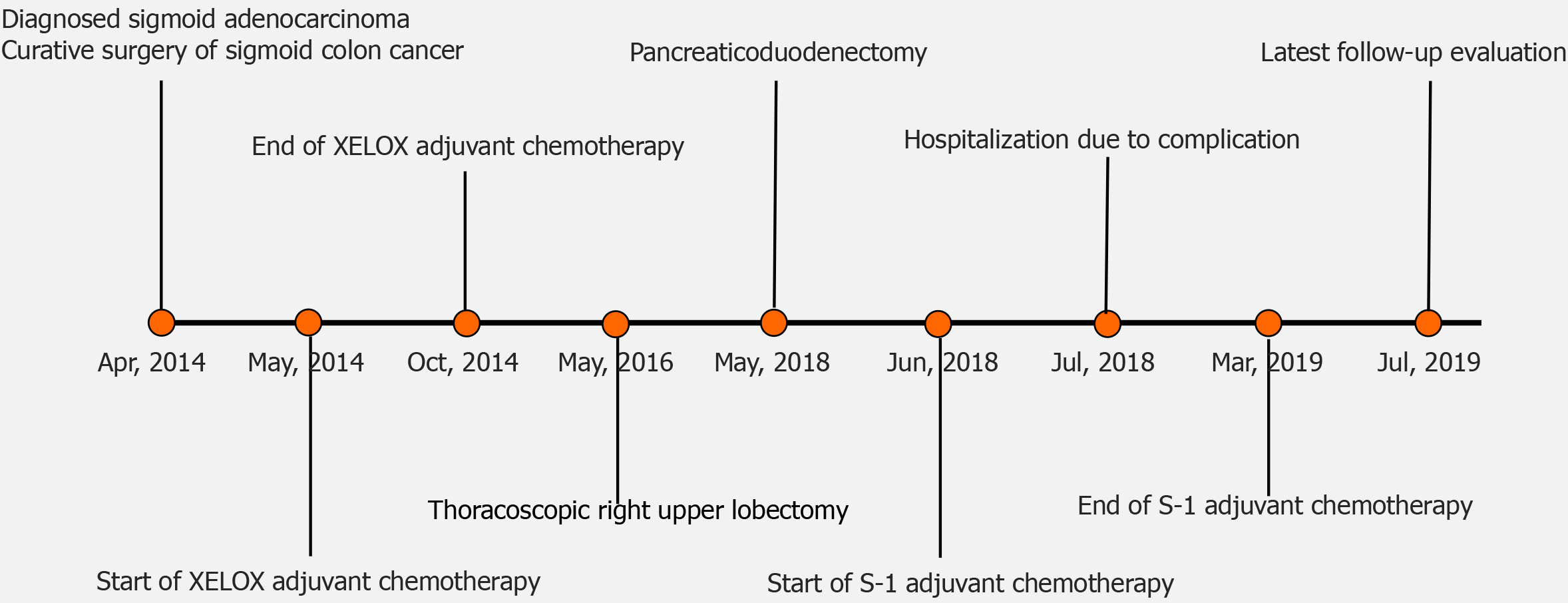
Frontiers | Feasibility Study of a Modified XELOX Adjuvant Chemotherapy for High-Recurrence Risk Patients With Operated Stage III Colon Cancer
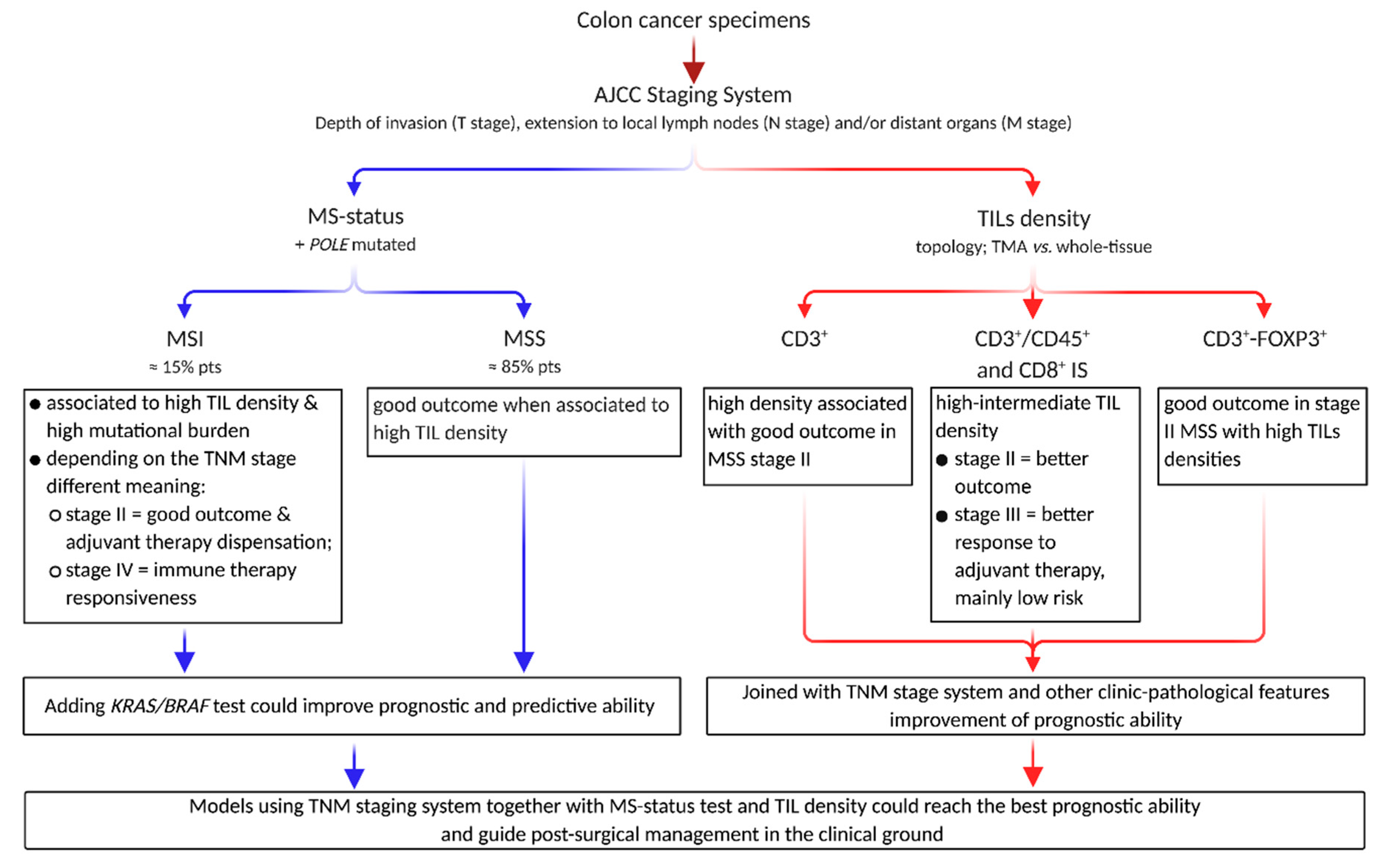
IJMS | Free Full-Text | Prognostic and Predictive Cross-Roads of Microsatellite Instability and Immune Response to Colon Cancer
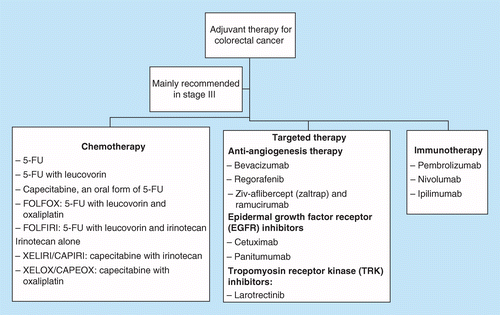
Oral adjuvant therapy for colorectal cancer: recent developments and future targets | Therapeutic Delivery

Table 1 from The efficacy of XELOX and FOLFOX adjuvant chemotherapy in stage III colorectal cancer patients with low preoperative serum albumin levels | Semantic Scholar

Impact of age and medical comorbidity on adjuvant treatment outcomes for stage III colon cancer: a pooled analysis of individual patient data from four randomized, controlled trials - ScienceDirect

3 versus 6 months of adjuvant oxaliplatin-fluoropyrimidine combination therapy for colorectal cancer (SCOT): an international, randomised, phase 3, non-inferiority trial - The Lancet Oncology

Duration of Oxaliplatin-Containing Adjuvant Therapy for Stage III Colon Cancer: ASCO Clinical Practice Guideline | Journal of Clinical Oncology

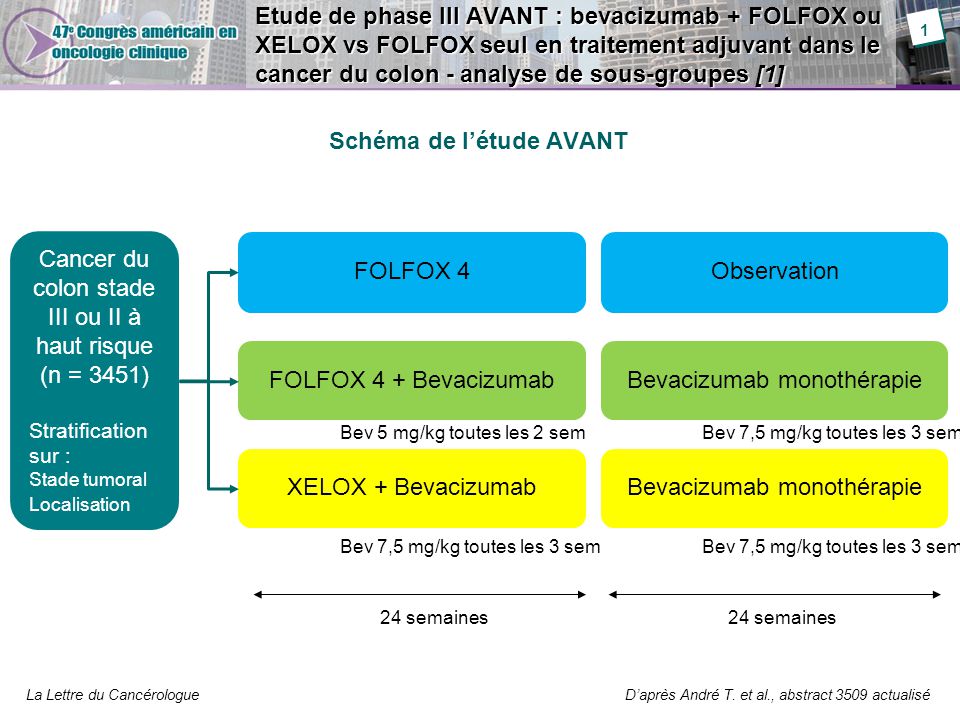
Bevacizumab (Bev) in combination with XELOX or FOLFOX-4: updated efficacy results from XELOX-1 / NO16966, a randomized phase III trial in first-line metastatic. - ppt video online download

Effect of adjuvant capecitabine or fluorouracil, with or without oxaliplatin, on survival outcomes in stage III colon cancer and the effect of oxaliplatin on post-relapse survival: a pooled analysis of individual patient

Effect of duration of adjuvant chemotherapy for patients with stage III colon cancer (IDEA collaboration): final results from a prospective, pooled analysis of six randomised, phase 3 trials - The Lancet Oncology